Robots and vision inspect pharmaceutical vials
Custom design combines with off-the-shelf components to detect tiny particles.
By Andrew Wilson, Editor
Time-sensitive drugs often must be administered within days or even hours of manufacturing. Before they are administered to the patient, these drugs must be manufactured (harvested), packaged, and inspected-a process that mandates checking for the presence of foreign particles in the drug after the manufacturing process. To do this, Gerald Budd, president of Phoenix Imaging, and his colleagues have developed a robotic-based vision system that can automatically load and unload products onto a test stand and inspect for such particles in a fraction of the time required by human operators.
Gerald Budd and Jules Knapp teamed up in 1996 to tackle this problem, and the work led to the creation of a better technique to characterize particles in clear solutions. The technology has collectively become known as the ParticleScope System (see Fig. 1).
“In the design of the system,” says Budd, “a number of important physical properties characterizing the fluid dynamics of the system and the motion of particles within the fluid needed to be considered.” These relate to the vessel size and shape, the fluid viscosity, surface tension, and the fluid fill volume, as well as the size, shape, and density of any particles that may be present. By tabulating all of these properties into a single matrix, Phoenix Imaging developed a specific velocity motion profile for numerous products.
“When each vial is rotated,” says Budd, “larger particles are spun via centripetal force to the outside of the vial while smaller particles (and those with less mass) will remain in the vial center. Thus, by properly controlling the velocity, it is possible to anticipate particle position and accurately measure the particle size.”
Fluid in motion
One important observation made by Budd and Knapp was the motion of the fluid in the vial as it rotated. The long-standing belief that centripetal force was the primary particle motivator and that once set in motion particles would stay near the interior wall was disproved. Budd explains, “During the initial period in which the container is spun, centripetal force moves the particles outward against the interior wall. However, shortly after the rotational motion has stopped, a vortex is created in the fluid, and particles of lower mass take on a spiral motion toward the axis of rotation within the vial. If the particle has sufficiently low mass the vortex will lift the particle off the floor of the container.”
This method allows particles to be separated further by analyzing the distance a specific size particle is suspended above the floor of the container at a particular period of time during the test. Smaller particles, being lighter, will rise higher in the vortex.
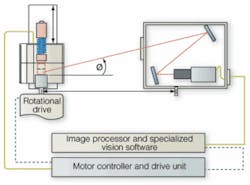
In the ParticleScope design, a number of illumination, imaging, motion, and robotics challenges had to be overcome. “To ideally illuminate the vial as it rotates,” says Budd, “you need to uniformly light the sample from all sides. However, because the system was required to image the sample as it rotated, Phoenix engineers developed a custom U-shaped LED light module using more than 1000 630-nm red LEDs from Nichia. Using a custom-built current controller, light intensity is controlled from the system’s host PC through standard digital I/O lines. “Thus,” says Budd, “low-opacity particles (such as fibers) can be illuminated more effectively using low light levels, while heavy (optically dense) particles can be lit with a brighter, more intense light.”
To image particles within each vial, the engineers realized that a large depth of field (DOF) would be required. “This was accomplished using a 100-mm-focal-length lens from Kowa and folded optics to increase the object-to-camera distance and thus the depth of field on the object side,” says Budd (see Fig. 2). At the same time, such folded optics allowed the space between the imaging system and the vial under test to be reduced (space is a premium on the pharmaceutical-manufacturing floor).
Says Budd, a number of other methods could have been used, including the use of wavefront encoding techniques from CDM Optics, now part of Omnivision Technologies (see Vision Systems Design, August 1998, p. 15), or the use of DOF enhancers such as those offered by Fluid Imaging (see Vision Systems Design, November 2003, p.27). “Both methods,” says Budd, “would have been more expensive to implement than the folded optics technique used in the ParticleScope System.”
In operation, reflected images from the vials are transmitted through the folded-optic assembly and imaged by a 1300 × 1030-pixel Camera Link M4+ camera from JAI Pulnix. Interfaced to a PC-based PIXCI CL2 64-bit/66-MHz PCI frame grabber from Epix, images are directly transferred to the host PC memory for image processing. “The motion control of the system needed to be tightly integrated with the imaging system,” says Budd.
To achieve this, a CMAX-810 CE stepper motor from Advanced Micro Systems was also interfaced to the PC over an RS-232 interface. One important component of the system involves a uniquely defined sequence for spinning the container (vial) called the Velocity Motion Profile (VMP). It is unique to each containers shape, size, fill level, viscosity, and even the surface tension of the fluid. It is critical that the energy transferred into the system does not distort the meniscus, or cavitation may result in a signal for false rejection of product.
The program for the proper VMP is downloaded to the stepper controller before the start of the sequence. This allows the program to operate very quickly with minimal interaction with the PC. “It may be possible to implement a servo motor in the application provided that it has enough torque to start and stop quickly, however, it would have impacted the cost of the system,” says Budd.
In its most basic form, the ParticleScope System can be hand-loaded by a technician. However, this can be a time-consuming operation if several hundred vials are to be inspected. Alternatively, the engineers decided to provide an option to load and unload vials automatically. They chose a small VP Series robot from Denso Robotics that could be mounted inverted above the laboratory unit. “Vials placed on the rotary actuator need to be positioned accurately to ensure that they are not damaged, and the Denso Robots have a repeatability of approximately 20 µm,” says Budd, “a figure that can easily be achieved using the VP Series.”
The ParticleScope System acquires multiple images in rapid succession; this ensures that if a particle is present in the container it will be detected. After images are captured they need to be processed very rapidly. To do this, the system currently uses a 3.6-GHz PC with a 1066-MHz front-side bus using fast dual-channel DDR2 memory running the Windows XP Pro operating system. “We are always watching for new developments in PC technology,” says Budd. “As soon as faster models are announced we benchmark them in using a series of in-house developed tests and, if they prove to be faster, integrate them into production systems.”
The most interesting part of the technology is that the system will compare the results of an image to other images in the sequence as part of the process for evaluating prominent dimensions. In fact, the ParticleScope system will measures the particle size at least six times before accurately reporting the particles equivalent diameter. Particle size can be accurately measured from 50 to 350 µm (±10 µm) and from 351 to 1000 µm (±25 µm) with near perfect linearity (R² = 0.998).
Get a GUI
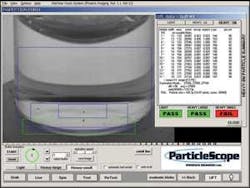
At the heart of the ParticleScope system is the GUI that controls the motion of the robot, the LED lighting module, motion of the rotary stage, and image-processing functions (see Fig. 3). Developed using Microsoft Visual Studio, the interface edits and selects the prewritten routines that include robot and motion control. “To control the motion of the robot,” says Budd, “we integrated Denso’s Wincaps II programming software as callable routines into the user interface. This allows the system to handle various size vials and vial spacing.”
Using this software allowed Phoenix Imaging to first view the robot’s actions in a 3-D simulation to determine obstacle clearances and verify reach and to develop and troubleshoot programs. “After the robot is operational, its actions can be monitored remotely by viewing nearly real-time 3-D simulation via RS-232C, Ethernet connections, and special software,” he says. Similarly, to control the motion of the rotary table, simple commands such as start, stop, read motion status, and force encoder position can be sent from over the PC’s RS-232 interface to the CMAX-810 microstep positioning controller.
To provide the image processing required in the system, Phoenix Imaging used its OEM vision package, the PC-based VisionMaker Development Package. “VisionMaker is composed of VisionMaker ToolBox and VisionMaker Algorithm Editor,” says Budd. VisionMaker ToolBox software is provided as Active-X controls with access to a large image-processing library. Each of the tools includes a vision preprocessor that reduces the visual information into a concise form that can be quickly processed.
To provide programming access using Active-X Controls, the VisionMaker ToolBox can be used with Microsoft Visual Studio for drag and drop “on-the-form” programming. “Because VisionMaker Tool Box VIEW.OCX and PITOOLS.OCX are custom controls for Visual Basic and Visual C++, they provide an extension to the Visual Basic ToolBox or the Visual C++ Application Studio Control Palette. In this way, we could add custom controls to the user interface,” says Budd (see Fig. 4).
Using blob-analysis tools combined with motion-analysis algorithms allows a number of parameters to be simultaneously monitored. These include the width, area, and gray-scale summation of specific particles in the vial. “Interestingly,” says Budd, “by computing a trajectory map of particles within the fluid, the system can visualize whether there are multiple particles present or whether the system has imaged one particle and its reflection from the bottom of the vial. This is an important image-analysis step because the reflected image is often distorted and appears much larger than the actual particle.”
Advantages and benefits
In the past, the inspection of particles within fluids was probabilistic in nature, one with human inspectors required to perform a statistical analysis on thousands of vials before a 1% confidence level in the batch quality could be determined. In today’s biopharmaceutical industry, where anticancer medications are manufactured in very small, expensive clinical-trial batches of less than 3000 vials, it is nearly impossible to approach the task with human inspectors.
“Worse,” says Budd, “such drugs may have a very short shelf life, and testing needs to be accomplished very rapidly. Because the ParticleScope is a deterministic method of measuring particles and not a probabilistic one, no such statistical measurements need to be made. “By analyzing just one sample,” says Budd, “we can determine the quality of the sample-no probabilistic batch methods are required.”
The system was unveiled to the pharmaceutical industry at the recent PDA Visual Inspection Forum (November 2005; Bethesda, MD , USA; www.pda.org/PDF/Meetings/05-visual_inspection.pdf). The technology is protected by multiple patents and is currently being reviewed by several pharmaceutical-equipment builders in an effort to implement it in high-speed (150-300 vials/min) production machines.
Understanding particle inspection
“Before any apparatus or automation can be implemented in the manufacturing process,” says Gerald Budd, Phoenix Imaging president, “the cGMP requirements of the US Food and Drug Administration mandate that the system be equivalent or better than the manual (human) based system that it replaces.” In the past, human operators performed the visual inspection for the presence of particulate counts in pharmaceutical products using a de facto standard statistical methodology developed by Julius Knapp, a pharmaceutical-industry expert in the field of particle inspection, and Lee Abramson. The technique required that a statistically significant number of samples be inspected to determine if the batch was of acceptable quality. The sampling inspection found its basis in military statistical methods formulated to test large quantities of vaccines during World War II.
The standard for rejected material was established as particulate contamination that could be detected by a human inspector at the clinical injection site. The fact that no “standard testing conditions” were established has left the industry with a purposely vague inspection requirement.
Phoenix Imaging has proposed a standard set of conditions to aid the global harmonization of the manual inspection procedures. First was the creation of a “standard environment” for human inspection of pharmaceutical products used for injection. The Manual Inspection Booth (MIB-100) provides a large volume of uniform illumination. The booth provides a 17-liter volume in the center portion that is uniformly lit to 550 foot candles ±5% using standard 5000 K Biax lamps from General Electric and a pair of precision power supplies with active-loop feedback. The illumination technique and construction of the MIB-100/200 are protected by several US and international design patents. The large working volume accommodates inspectors of just about any size by allowing shelves and armrest to be adjusted without the use of any tools. The appliances are adjusted so that the extended arm with elbow resting on pad is positioned in the center of the uniform illumination volume. A PLC ensures that the time is consistent for each container inspection.
Next, the “baseline” of the human inspectors is determined. The baseline is the particle size that human inspectors are capable of detecting 70.71% of the time, as established by the Knapp-Abramson technique. The baseline will be used to establish the minimum size particle that must be detected to reject a product. The problem was solved with the development of a “Standard Calibration Set” consisting of known particle sizes. Phoenix Imaging includes particles much smaller and much larger that could be detected by human inspectors using the MIB.
To perform this test accurately, a standard particle set of US National Institute of Standards and Technology-traceable stainless-steel and glass microspheres ranging in diameter from 50 to 1000 µm are placed in a known amount of ultraclean water known as WFI (water for injection). Two different particle materials are selected to determine if opacity was a factor in the detection level.
Automated approaches
A number of companies developed automated inspection techniques to handle larger volumes of products. As the techniques evolved, it was determined that particles must be put in motion to detect their presence. The first commercially viable systems were created using a technique that would rotate the container, usually a glass vial or ampule of less than 5 ml, and then stop the rotation quickly. The fluid would begin to rotate in the same direction as the container, and it was believed that any contaminating particles would move with the fluid.
Says Budd, “The initial system approach would implement a linescan detector on one side of the container and a collimated light source on the opposite side of the container. The particles moving with the fluid would pass between the light source and the detector, providing a pulse whose height was proportional to the size of the particle. Unfortunately, because the bottom of the typical small vial is inverted, the imager could not inspect the bottom where particles with a diameter larger than ~150 µm would settle in less than 250 ms. To overcome this problem, many companies decided to spin each vial at a higher rotational velocity. While this raises any particulate matter higher in the vial, the resulting increase in energy deforms the meniscus shape and causes cavitations (production of tiny air bubbles) that are detected as defects.”
Several European companies produce automated systems for this task. “In these systems,” says Budd, “LED backlights are used, and the vial is inspected orthogonally using an area-array camera or a linescan sensor.” These systems are often based on the principle of light extinction, in which the defect is represented as a binary object. “Should a cubic particle be aligned orthogonally to the source, light may not be attenuated below the threshold level or may be reflected toward the detector and be misinterpreted by the system,” he says. - AW
Company Info
Advanced Micro Systems
Nashua, NH, USA
www.ams2000.com
CDM Optics (now part of Omnivision Technologies)
Sunnyvale, CA, USA
www.ovt.com
Denso Robotics
Long Beach, CA, USA
www.densorobotics.com
Epix
Buffalo Grove, IL, USA
www.epixinc.com
Fluid Imaging
Edgecomb, ME, USA
www.fluidimaging.com
Food and Drug Administration
Rockville, MD, USA
www.fda.gov
General Electric
www.gelighting.com
JAI Pulnix
San Jose, CA, USA
www.pulnix.com
Kowa
Tokyo, Japan
www.kowa.co.jp
National Institute of Standards and Technology
Gaithersburg, MD, USA
www.nist.gov
Nichia
Tokushima, Japan
www.nichia.com
Phoenix Imaging
Livonia, MI, USA
www.phoeniximaging.com