Vision system validates medical containers
By Graham Sprigg,Contributing Editor
Precise reading of labels and characters on pharmaceutical containers mandates the use of automated vision-inspection systems.
The pharmaceutical industry relies heavily on automated process-control and quality-assurance systems to ensure that batch production is carried out repeatably, reliably, and accurately. Like most manufacturing industries, especially those concerned with the production of high-value end products, pharmaceutical producers are constantly looking at ways to increase throughput, maximise yields, and improve the flexibility of batch manufacturing. Moreover, there is probably no other industry where proper quality assurance and quality control can literally make the difference between cure and casualty.
Whereas the correct control of production processes, including product labeling, traceability of raw materials, and tracking of finished products, is important in other industries (such as food and beverage production), the market drivers differ for pharmaceutical containers. In many countries, government legislation largely governs the processes by which pharmaceutical manufacturers produce products, and the resulting data are used to verify the quality of their products. Consequently, all automation and inspection technologies introduced to a production plant must meet the strict validation criteria that govern the industry. In many inspection applications, machine-vision systems are the obvious choice for pharmaceutical manufacturers who need 100% inspection. In addition, these systems must comply with US Food and Drug Administration (FDA) regulations.
As a result, vision inspection systems also are being applied to incoming materials, particularly for the proofreading of information provided by raw-material suppliers. In practice, incoming materials inspection has been fraught with problems and is responsible for more product recalls than any other aspect of pharmaceutical production. Gary Parish, president of GSMA/Parish Automation (Palm Bay, FL), a supplier of machine-vision systems to the pharmaceutical industry, says, "A single hyphen missing from a label can change the directions for usage from 'take 1-2 tablets daily' to 'take 1 2 tablets daily. Picking up this type of error places a big burden on incoming inspection departments."
Production line mixups can have serious user and legal consequences. Incoming materials are therefore meticulously inspected, especially packaging data, for errors.
To meet FDA Good Manufacturing Practices (GMPs) guidelines, all pharmaceutical manufacturers have established departmental procedures to make sure that incoming materials, such as labels, inserts, and cartons, carry information that exactly matches the information originally approved by the manufacturer. Often called a golden or master copy, a signed original is kept on file to compare against newly printed incoming materials. But rather than making inspection easier, these masters add to the proofreading burden, as many originals do not contain the same information as the incoming material receipt. Many receipts are photocopies, printouts, or color-coded.
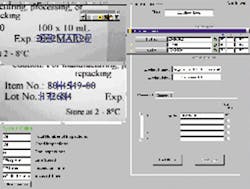
FIGURE 1. Xyntek MVX-OCV inspection system uses the Cognex MVS-8000 frame grabber and software for image acquisition, which is controlled using a graphical user interface. Inspection application runs on a Windows NT 4.0 PC platform. A programmable logic controller (PLC) triggers the camera to acquire an image, which is then analyzed to determine whether or not the characters match those of the trained system. Pass/fail results are then communicated back to the PLC, which keeps track of each result, applies a label, and controls the destination of each bottle.
The problem is compounded by the existence of thousands of SKUs (stock-keeping units), product variations, and different language versions, which all complicate the inspection task. Although high-speed, on-line inspection is improving, thanks to the use of barcodes and computer vision systems, incoming inspection of text and graphics copy is still often done by human inspectors who compare the master with the sample.
Proofreading systemsSome companies have even added extra inspection tasks to ensure product quality. In addition to the GMPs that drug companies must follow, AstraZeneca (Wilmington, DE) has added a set of US military standards for passing or failing rules for the inspection of incoming lots of materials. The military standard, in this case Rule 105-E, establishes tight guidelines for inspection that AstraZeneca applies to its labels, cartons, inserts, and containers.For example, the company generally starts with a sample lot of 800 inserts, because the 100% inspection of a lot of 150,000 inserts is not feasible. Defects on an insert with text and graphics are classified into different critical levels for rejecting the entire lot of 150,000 inserts. They include nontolerance, such as incorrect name, product, or barcode. Any one defective sample rejects the entire lot; critical, such as unreadable legal copy or blank areas of legal copy. Defects in 0.65% of the 800 samples can reject the entire lot; major defects, such as color out of specification or torn, wrinkled, or blurred copy. Defects in 1.0% of the 800 samples can reject the entire lot; and minor defects, such as off-register, missing (noncritical) print, or misspellings. Errors in 4.0% of the 800 samples can reject the entire lot. In practice, the company has found that automated vision inspection improves accuracy and reduces inspection time by 70%.
To handle these kinds of inspection challenges, in the early 1990s GSMA's sister company Parish Automation Inc. (Palm Bay, FL) introduced the first electronic image comparator (proofreader) for the pharmaceutical industry. Called Inspec, the system worked by having an acceptable label, leaflet, or carton placed under a CCD camera and capturing an image in memory using a Coreco Imaging (Bedford, MA) vision board, a predecessor to the current PCVision PCI-bus frame grabber. The image was sent to a computer processor that produced an electronic image negative. This image was then used as a master reference image that was subtracted from the incoming sample. Any image differences would be highlighted and "flashed" on a monitor for operator viewing and grading.
The real challenge, according to Gary Parish, is that "many proofreaders are under the impression that they are as accurate and faster than electronic inspection. The best way to demonstrate the problems that human inspectors confront is to ask the proofreader to take a 'spot the 10 differences' drawing and compare the inspectors' performance with that of a vision computer. The proofreaders usually take several minutes, if they are able to find the differences, while the vision-based computer can highlight the differences in less than one second."
Materials come in all sizes, so the manual inspection approach requires an operator to either step the camera or material through each level of comparison. The area of inspection or field of view (FOV) of the CCD camera is based on the size and complexity of the material being inspected. If the operator is inspecting a small label with Japanese copy, a small FOV may be needed to pick up a missing character stroke. The inspection area could occupy less than 1 x 1 in. of the material. Since the CCD camera is based on video technology, it generally provides a 659 x 494-pixel image. Higher pixel densities translate into higher accuracies.
FIGURE 2. In the Xyntek MVX-OCV system, inspection setup uses a graphical user interface. First, an image of the codes to be inspected is acquired and displayed on the configuration screen. Second, each character is "trained" using the train fonts screen (top). Using a CCD camera, the system is trained to recognize up to 10 different font sets and verify the printout of various sizes or typefaces. Third, using a define-strings screen, the text position, search area, character angle, and spacing on the label are configured. In some applications, two cameras are used to ensure that correct multiple codes are printed on a label (bottom).
To take incoming materials inspection to a higher level, the addition of a fully automated x-y-coordinate robot steps the camera over the entire insert area, and inspection tasks are performed at the required resolution. The latest Inspec systems are GSMA's RoboSpect and PageSpect. These vision systems combine robotics with machine-vision technology to reduce inspection times by up to 95%. RobotSpect automatically inspects printed press sheets up to 40 x 60 in. in area for the pharmaceutical industry. PageSpect is a tabletop version for inspecting materials up to 12 x 18 in., as well as roll or pin-fed labels.
To meet strict validation requirements, both systems control the entire inspection process. In addition to the Coreco Imaging PCVision frame grabber, which captures and transfers images to the host processor in less than 4 µs, GSMA has added several automated features, including a programmable x-y robotic inspection stage, one or two cameras with different resolutions, and controlled lighting. The current versions of RoboSpect and PageSpect can inspect up to 60 labels per minute. An entire 8 x 11-in. insert can be inspected in less than one minute, depending upon the amount of copy on the page.
Optical charactersIn addition to labels, many pharmaceutical companies want to verify every character on a medical container. Legislation is again a major driver for character verification, with the FDA's 21 CFR Part II regulation taking effect. This character verification relates to electronic records that are now considered to be equivalent to paper records and electronic signatures. The regulation validates an electronic signature as the equivalent of a handwritten signoff.To help pharmaceutical manufacturers comply with this regulation, an optical-character-verification (OCV) system has been developed as a result of an alliance between Cognex Corp. (Natick, MA) and Xyntek Inc. (Yardley, PA). Xyntek developed the MVX-OCV system using the Cognex MVS-8000 frame grabber as the platform for the vision inspection system (see Fig. 1). Full-frame image acquisition is controlled using a graphical user interface (GUI) with data management and control capabilities. The application runs on a Windows NT 4.0 PC platform. The system relies on Cognex vision algorithms to perform OCV. With this approach, Xyntek has integrated a packaged vision platform for the pharmaceutical industry that provides off-the-shelf OCV.
The pharmaceutical companies using the MVX-OCV system are looking primarily at date and lot codes on product labels that are subsequently adhered to bottles. Inspection times typically run at 300 to 600 labels per minute, and verification takes 1 ms/character.
The GUI configures the inspection process in three steps. First, an image of the codes to be inspected is acquired and displayed on the configuration screen. Second, each character is "trained" using a train fonts screen (see Fig. 2). Each camera is capable of being trained to recognize up to 10 different font sets, and the system can be trained to recognize and verify print of various sizes or typefaces. Third, using a define-strings screen, the text position, search area, character angle, and spacing on the label are configured. During operation, an image of each product label is captured, and the data are verified to ensure that the correct codes are printed on each label.
For example, a pharmaceutical company required 100% print verification of labels used on bottles containing blood-glucose test strips. The complex 84-character labels had to be inspected at 120 bottles per minute. The company installed an MVX-OCV system that performs as many as 150,000 label inspections a day while keeping the false reject rate below 1%, per customer requirements. The labels, which measure 2 x 4 in., are mounted on the bottles by a continuous horizontal web. These labels are indexed individually in front of a side-mounted camera.
As the label enters the camera's FOV, a programmable logic controller (PLC) triggers the camera to acquire an image. This image is then analyzed to determine whether or not the characters match those of the trained label. Pass/fail results are then communicated back to the PLC, which keeps track of each result, applies a label, and controls the destination of each bottle. Bottles with acceptable labels are sent further downstream to a final packing station. Defectively labeled bottles are pneumatically ejected from the line. After the faulty label is removed, these bottles are then relabeled and reinspected.
The Xyntek MVX-OCV system provides 100% inspection, character by character, as well as a running log of faults. The log is used to print out statistics to determine the cause of failed labels. System options permit the display of a real-time image or a frozen image of the last failed label.
Liquid particle inspectionAutomatic vision-inspection systems for the pharmaceutical industry have also been developed that can detect particles within liquids, as well as such container defects as wrong dimensions, cosmetic flaws, and blemishes, among others. These systems can inspect such containers as vials, ampoules, and syringes at speeds to 400 products per minute.In liquid-particle-inspection applications, pharmaceutical products are generally inspected by passing the products through several inspection stations interconnected to an overall vision system, mostly using an image-subtraction method. In this approach, containers filled with liquid are rotated at high speed. When the rotation of the container is stopped, the liquid and any dispersed particles continue to move. The vision-system camera, which rotates in synchronization with the containers, is triggered to acquire a sequence of images. These images are captured and delivered to the system image processor. There, the images are compared to each other in an overlay image, pixel by pixel.
Particles that change location during the image sequence (that is, move within the liquid) are identified as defects. Dust or light scratches on the container surface do not change their position between images and are therefore not graded as defects in this inspection method.
Using two or three particle-inspection stations to cover a moving container line allows the variation of rotation speed to detect light or heavy particles. The set points for rotation speed and stop-and-inspect are stored together with the product inspection configuration (recipe) for each product. Positioning drives are used to adjust the rotation modules, and various sensors check for proper alignment and rotation speed.
Depending on product range and container type, different illumination techniques are used to highlight the inspection area. In the light-reflection method, concentrated light is sent through the bottom surface of the container. The inspection camera, which is positioned to look through the side surfaces of the container, detects the light reflections scattered by particles against a dark background.
In the light-transmission method, light is projected through the side surface of the container. The inspection camera, which is positioned to look through the other side of the container, picks up the shadows created by any particles. Heavy nonmoving-type particles on the inside bottom surface of the container require special lighting techniques.
Company InformationAstraZenecaWilmington, DE 19850Web: www.astra-zeneca.comCognex Corp.
Natick, MA 01760
Web: www.cognex.com
Coreco Imaging
Bedford, MA 01730
Web: www.coreco.com
GSMA/Parish Automation
Palm Bay, FL 32905
Web: www.gsma.com
Xyntek Inc.
Yardley, PA 19067
Web: www.xyntekinc.com