Image processing speeds DNA damage tests
Advanced image-processing system automates and accelerates the detection of irregular samples of DNA.
By Mark Lockie, Contributing Editor
At the Gray Cancer Institute (Northwood, Middlesex, UK)—a UK-funded cancer-research institute—the advanced-technology-development (ATD) group is working on methods for looking at the damage to DNA caused when it has undergone certain treatments. The group is combining advanced microscopes, cameras, several image-processing stages, and original software to achieve a high level of automation. One method it uses is called single-cell gel electrophoresis, or 'comet assay,' originally developed by researchers Östling and Johanson of the Gustaf Werner Institute (Uppsala University; Uppsala, Sweden).
Comet assay is one of the fastest tools available for assessing DNA damage and has far-reaching implications, both in cancer research and elsewhere. For example, by using comet assay, scientists can measure the radio-sensitivity of tumors in patients before deciding on treatment and dose prescription.
Says professor Borivoj Vojnovic, ATD group head, "For our purposes we use cultured tumor cells. These are fixed in a gel, such as agarose, and are subjected to a small electrical field in the region of 1 V/cm. This has the effect of teasing out any broken DNA fragments, which then migrate through the tiny pores in the gel, creating a comet-shaped tail. The smaller the fragments, the farther they travel, and the worse the damage to the DNA."
The comets are viewed under fluorescent conditions. In an undamaged cell, the glow from the fluorescent marker is localized at the nucleus, with the amount of glow given off proportional to the amount of DNA contained. The assessment of individual comets is generally a labor-intensive process, especially considering that every prepared slide contains approximately 1000 comets. However, integrating microscopes, cameras, and image-processing stages is resulting in a higher level of automation.
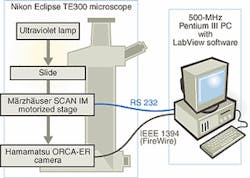
FIGURE 2. In fully automated comet-assay system, image manipulation is not needed due to the high dynamic range of the Hamamatsu 12-bit ORCA-ER camera used to capture the comet image. The cooled CCD camera provides an extended range of 1344 X 1024 pixels and full remote control from the connected PC via an IEEE 1394 (FireWire) interface. Comet location and focusing are performed by an inverted Nikon Eclipse TE300 microscope using a motorized Märzhäuser SCAN IM 120 X 100 microscope stage. Feedback from the PC triggers the microscope lens to alter its position before a new image is captured. This iterative procedure continues until a highly focused image is captured.
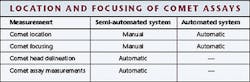
The ATD group has developed one operational semi-automated comet assay system and is in the process of developing a fully automated system. In the semi-automated system, the technician must position each individual comet assay within the center of the microscope's field of view and put it into focus. The remainder of the process is automated, however, and results in an analysis rate of approximately 200 to 300 comets/hour. In the fully automated system, the location and the focusing of comets are automatically performed, but subsequent processing must be integrated (see table and Fig. 1).
Because comet assay heads are extremely bright compared with their surrounding tail, a camera with a high dynamic range is required. The semi-automated system takes two images at different integration times because the camera can only capture 8-bit images. A short integration time captures the most intense image detail possible without overloading the CCD sensor, and an additional image is then captured at an increased integration time. These images are digitally combined to create another image of increased dynamic range by replacing the saturated pixels from the second image with the appropriately scaled information from the first image. This method allows the analysis of comets with extremely weak tails compared with the strong intensity of the comet head.
In the fully automated system, this image manipulation is not needed due to the higher dynamic range of the Hamamatsu (Bridgewater, NJ) 12-bit ORCA-ER camera used to capture the comet image (see Fig. 2). This cooled CCD camera provides an extended range of 1344 X 1024 pixels and full remote control from the connected PC via an IEEE 1394 (FireWire) interface. In addition to obtaining wider dynamic range, the ATD group chose FireWire because it eliminates the need for a frame grabber board in the PC.
The processes of comet location and focusing are performed in the fully automated system by an inverted Nikon Eclipse TE300 microscope using a motorized SCAN IM 120 X 100 microscope stage from Märzhäuser (Wetzlar-Steindorf, Germany). This setup is used to sequentially scan the slide in steps while the images are captured. Captured images are sent to the PC, and a focus indicator is then produced using a fast-Fourier-transform approach. Feedback from the PC triggers the microscope lens to alter its position before a new image is captured. This iterative procedure continues until a highly focused image is captured.
An interesting processing point at this autofocusing and positioning step is the ability of the camera to bin pixels. The camera takes each 4 X 4-pixel group and reduces it to a single pixel. This step decreases the image resolution before sending it across the interface but increases the transmission and processing speed by a factor of approximately 16. After the optimum point is reached, however, the final image is taken at the full resolution for subsequent processing.
The software development tool used in both systems is National Instruments LabWindows/CVI, which is part of Measurement Studio. This C-based software is designed for developing measurement applications and allows the integration of image capture, processing, and data presentation in one package.
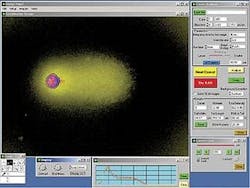
Image processingHaving acquired a focused comet image, the PC-based image-processing software automatically removes the background fluorescence and places rectangular region-of-interest boxes around each comet in the field of view by thresholding and prior knowledge about approximate comet size (see Fig. 3). In the semi-automated system the most central comet is selected for processing, but in future systems every comet in the field of view will be individually processed.The next step is to detect the position of the comet head and delineate it from the region of interest. Unfortunately, the type of cell lines that the ATD group wanted to measure were oddly shaped and had highly structured nuclei, thanks to clumping of the DNA. This condition meant that existing methodologies to delineate the comet head were not satisfactory, and a significant amount of original research was needed to get the system working effectively.
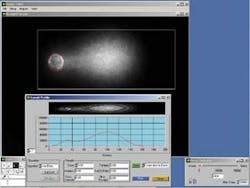
An algorithm developed by ATD group scientist Paul Barber uses a compact Hough transform in the formation of a radial map of the comet head (see Fig. 4). The algorithm—the Charm algorithm—finds the center of the nucleus by locating the pixel with the greatest response to the compact Hough transform. It then performs radial searching to determine the nucleus' most likely boundary.
Results of interestAfter the Charm algorithm has identified the comet head boundary, the software calculates measurements of interest to cancer researchers. These various measured parameters are outputted via dynamic data exchange to an Excel spreadsheet. ATD group scientific assistant Rosalind Locke comments, "All the measurements correlate in some way to the level of damage to the DNA. These include the comet moment, the percentage of DNA in the tail and the head, and the length of the tail." Automated image processing clearly plays an important role in this entire process. But even better results are expected, once the fully automated system is finished, both in terms of accuracy and speed.Biologist Peter Johnston in the Gray Cancer Institute experimental-oncology group says, "Comet assay provides a technique to examine the induction and repair of DNA damage in individual cells that, in turn, allows the discrimination of sensitive/resistant subpopulations. Automation will significantly improve the sensitivity of the assay and allow detection of damage at levels approaching those encountered during environmental, therapeutic, or accidental exposure to DNA damaging agents, such as ionizing radiation and other mutagens."
The world record for manually locating, focusing on, and capturing comet images is several hundred comets per hour. At the institute, a level of 200 to 300/hour is normal. With the new fully automated system the aim is to increase this speed to 10,000 per hour. "This will give researchers the luxury of performing more experiments or just give them extra thinking time," ATD group head Vojnovic says.
Company InformationCohu Inc.San Diego, CA 92186www.cohu.comGray Cancer InstituteNorthwood, Middlesex HA6 2JR, UKwww.graylab.ac.ukHamamatsu PhotonicsBridgewater, NJ 08807www.hamamatsu.comMärzhäuserWetzlar-Steindorf 35579, Germanywww.marzhauser.comNational InstrumentsAustin, TX 78759www.ni.comNikon Instruments Inc.Melville, NY 11747www.nikon.comUppsala UniversityUppsala, Swedenwww.uu.se